A recent study by Vanessa Li and colleagues, published in PNAS and carried out at the Montreal Neurological Institute-Hospital at McGill University, has revisited the early formation of visual system maps in the Xenopus optic tectum, using state-of-the-art optogenetic technologies to follow the emergence of retinotopic maps at subcellular resolution in the brains of living, developing Xenopus tadpoles.
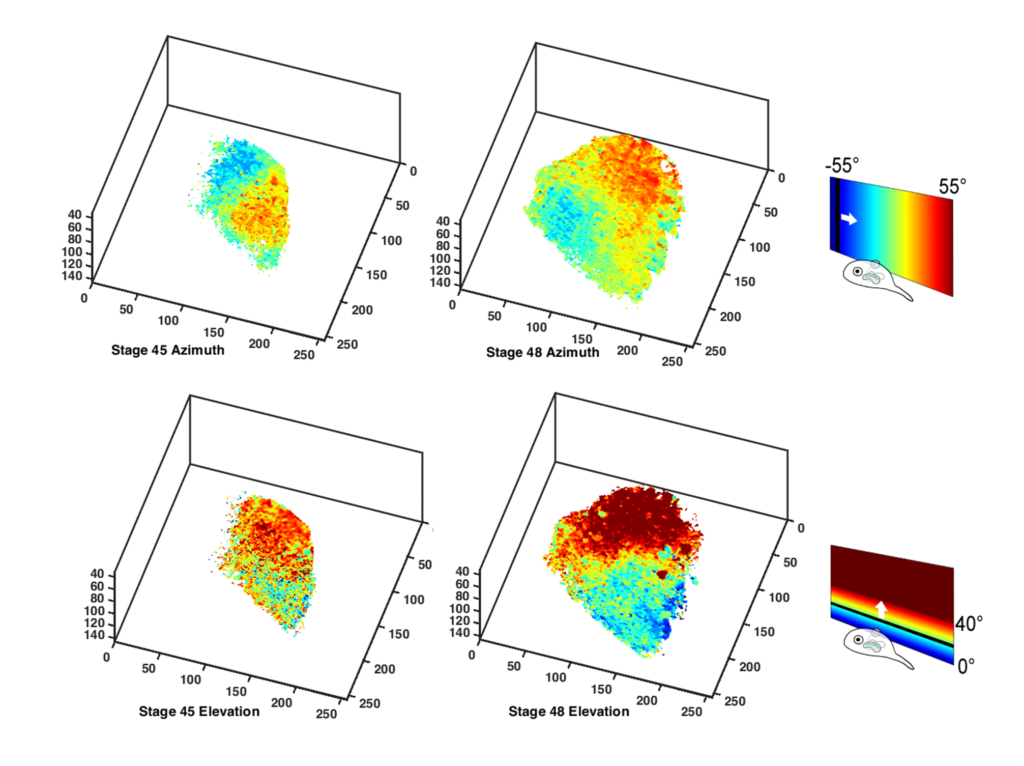
The researchers grew Xenopus tadpoles whose neurons were made to express the fluorescent protein GCaMP6s, which increases its brightness when neurons are activated. They then imaged their brains non-invasively using a high-speed 2-photon microscope, which can create three-dimensional reconstructions of the entire brain, akin to the brain sectioning performed in an MRI scan, but at much finer resolution. The researchers presented visual stimuli to the tadpoles and asked what cells in their brains had been activated by stimulation in different parts of the visual field. Using this data, they were able to generate cellular resolution retinotopic maps in the developing brain starting as early as just one day after the projections from the eye first arrived in the brain and following them in the same animals as they developed over subsequent days.
Their paper, published in the February 14 issue of Proceedings of the National Academy of Sciences, confirms researchers’ long-held notion that maps in the brain become increasingly precise over time as the brain grows, but also reveals some unexpected twists. One surprising finding was how remarkably well-organized certain aspects of the visual maps in the brain are already at their initial emergence. Another impressive finding was the observation that the retinotopic maps physically rotate within the developing brain over several days. These results highlight just how plastic and malleable the young brain can be, even despite the presence of chemoaffinity cues.
Research that confirms and expands on the discoveries of neuroscience pioneers Wilder Penfield and Roger Sperry
Just under a century ago the great neurosurgeon Dr. Wilder Penfield, founder of the Montreal Neurological Institute and Hospital, pioneered the treatment of epilepsy by surgical excision of the seizure focus from the cerebral cortex of patients. While carrying out these surgeries, which came to be known as the “Montreal procedure”, Penfield and his fellow neurosurgeons attempted to minimize damage by systematically stimulating the cortex to map out sites surrounding the presumptive lesion. By this process of systematic functional mapping of the cortex, Penfield discovered the orderly representations of sensory inputs and motor outputs that are mapped across the cortical surface. This discovery gave birth to the so-called sensory and motor homunculi, distorted “little men” representing the relative cortical territory allotted to each body part, something which is familiar to any student of neuroscience today.
Around the same time, future Nobel laureate Roger Sperry, still a young postdoctoral researcher, had become fascinated by the formation of sensory maps in the visual system. Sperry used a number of different species, including the African claw-toed frog Xenopus laevis, to examine how regenerating axonal projections from the retina to the primary visual center in the amphibian brain, called the optic tectum, achieved their remarkably precise organization in the target. Neurons from across the retina extend long axonal projections through the optic nerve to the brain where they remarkably reorganize themselves back into the same relative layout they had within the eye, creating a “retinotopic” map that effectively recreates within the brain the visual scene viewed by the eye. In a series of famous experiments Sperry and co-workers lesioned parts of the eye, even going so far as to rotate eyes by 180 degrees, to demonstrate that retinal axons find their destinations using prespecified molecular address codes that he referred to as “chemoaffinity” cues.
The research published today by Vanessa Li, Anne Schohl and Edward Ruthazer provides validation for these historic findings, and further expands our understanding of brain development and how adaptable it can be.







